Advice for the new intranasal vaccine, concerning stats from last year and more has been released.
You all know what time it is. Summer has officially ended, and winter preparedness season is beginning.
The Australian Technical Advisory Group on Immunisation (ATAGI) has released its official advice for administering 2026 influenza vaccines, with a strong (but unsurprising) emphasis on boosting vaccination rates.
This focus was mirrored by Professor Michael Kidd, the chief medical officer for Australia.
“Your recommendation to your patients is one of the most influential drivers of vaccination acceptance,” he wrote to GPs in an open letter.
“Research shows that a strong, confident endorsement from a trusted health professional makes a significant impact on patient decision-making.”
He noted some very minor improvements in flu vaccine uptake from 2024 to 2025, but a concerning drop among Aboriginal and Torres Strait Islander people from 22.7% to 22.4%.
“Uptake in children under 5 increased modestly from 25.8% in 2024 to 26.1% in 2025, yet remains significantly lower than the 31.9% achieved in 2022,” he wrote.
Related
“For adults aged 65 and older, uptake rose from 61% in 2024 to 61.1% in 2025, still notably below the 68.3% observed in 2022.”
According to the National Notifiable Disease Surveillance System, there have been 18,973 laboratory-confirmed cases of influenza as of 2 March, 2026.
“Your continued efforts are essential to improving uptake, particularly among children under 5, pregnant women, Aboriginal and Torres Strait Islander people, and adults aged 65 and older,” Professor Kidd said.
He also reinforced the safety of co-administering flu shots with any covid, RSV or other routine vaccine.
Influenza vaccination is recommended from six months of age, with two doses to be administered four weeks apart for a patient’s first flu immunisation. All vaccines available in Australia this year are trivalent.
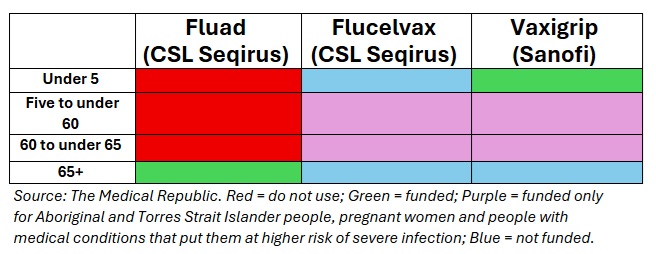
NIP funding vaccines:
NIP vaccines will be available to order in April 2026.
Fluad (NIP funded) or Fluzone High-Dose (private, Sanofi) are both equally preferred over standard influenza vaccines for those aged 65 years and over.
Flumist (AstraZeneca), the intranasal live attenuated vaccine, is available for the first time this year. It is approved for children aged two to 17 years and is available privately or through some state-based funding programs.
In New South Wales, Queensland and South Australia, flumist is free for children aged two years to under five years. In Western Australia, it is free for children aged two to under 12 years.
Other states do not have funding in place for flumist. It is as effective as inactivated influenza vaccines but is contraindicated for those with moderate or severe immunocompromise.





