Landmark review confirms relationship between gut dysbiosis and brain health and suggests Mediterranean diet could cut Alzheimer's risk by nearly a third.
Microbiome imbalances become progressively worse as cognitive decline advances, according to an international meta-analysis and dietary changes seem particularly effective when compared to other microbiota-targeted interventions.
Researchers from the George Washington University School of Medicine and Health Sciences in the United States performed the most thorough review of human studies to date on the link between dysbiosis and the development and progression of mild cognitive impairment (MCI) and Alzheimer’s disease (AD).
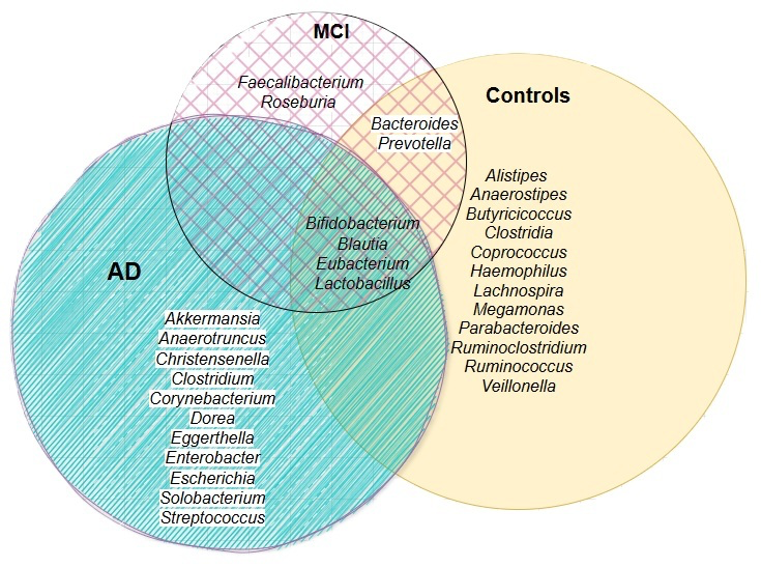
The typical pattern seen in the analysis was increased bacteria linked with inflammation, particularly Pseudomonadota and Actinomycetota, and reduced levels of beneficial bacteria, such as Faecalibacterium and Roseburia, when compared to healthy adults.
These beneficial bacteria normally produce short-chain fatty acids (SCFAs) that help reduce inflammation and support brain health.
These changes in the microbiome were more consistent and pronounced in AD than MCI, the researchers reported.
AD patients frequently had higher levels of Escherichia, Akkermansia, Enterobacter and Streptococcus, while healthy individuals had more beneficial bacteria, such as Ruminococcus, Parabacteroides and Coprococcus.
Some studies also observed functional changes in gut bacteria in those with AD, with reductions in energy metabolism, immune function, environmental adaptation and production of protective metabolites for brain health.
“These microbial signatures offer an important starting point for understanding how gut health may influence neuroinflammation and cognitive decline,” said lead researcher Associate Professor Leigh Frame of the George Washington University School of Medicine and Health Sciences.
There was some overlap between MCI and healthy controls in gut bacterium, as well as an overlap between AD, MCI, and healthy controls.
However, there were no genera uniquely shared between AD and healthy controls, or between AD and MCI.
The review, published in the Journal of the Alzheimer’s Association earlier this year, included 58 studies from the US, China, Netherlands and many more. Around half of the studies were case controlled, alongside nine systematic reviews, eight randomised-controlled trials (RCTs), three meta-analyses, three prospective cohorts, two cross-sectional, two Mendelian randomisations, one pre/post design of an exploratory intervention with no control group and one cohort and case-control mix.
The number of participants in the pooled analysis was not included.
Several of the RCTs included in the analysis tested microbiome interventions, finding that a Mediterranean-style diet was associated with higher levels of beneficial bacteria and as much as 32% lower risk of AD.
Many studies showed small improvements in cognitive scores, particularly with strains such as Bifidobacterium longum and Lactobacillus plantarum. However, these results were inconsistent, and the studies were mostly small and short term (12 weeks or less).
Although the review did not establish cause and effect, the findings aligned with emerging evidence of the impacts of gut dysbiosis on inflammation and brain health.
Related
Another recent review, published in Nutrition Research, found that microbiota-targeted interventions such as diets, probiotics and faecal microbiota transplantation (FMT), may have beneficial effects on cognitive function, particularly in individuals with MCI or early-stage AD.
Fifteen trials were analysed, involving 4275 participants aged over 45 years diagnosed with dementia, cognitive impairment or comorbidities associated with increased risk of neurocognitive disorders.
The review included RCTs and quasi-experimental studies published in peer-reviewed journals between 2012 and 2025 across Spain, China, the US, South Korea, Finland, Australia, Iran, Norway, Sweden and the Netherlands, with follow-up periods ranging from eight weeks to more than six years.
Cognitive outcomes across the studies were evaluated using validated tools such as the Mini-Mental State Examination (MMSE), the Montreal Cognitive Assessment (MoCA) and the Repeatable Battery for the Assessment of Neuropsychological Status (RBANS).
Overall, the analysis indicated that microbiota modulation was associated with improvements in memory, executive function and overall cognitive performance, which were particularly evident amongst individuals with prodromal cognitive impairment or MCI.
However, those with advanced AD showed limited response to microbiota-targeted interventions.
Across the studies, improvements in cognition were frequently associated with increased microbial diversity, enhanced production of SCFAs and reductions in neuroinflammatory markers.
“Overall, gut microbiota modulation represents a promising nonpharmacological strategy to support cognitive health, with early intervention appearing crucial for optimal benefit,” the authors wrote.
Dietary interventions appeared moderately effective, particularly for early cognitive decline, and had the strongest clinical evidence due to larger trials than other interventions.
Both the Mediterranean diet and the ketogenic diet were associated with improved cognitive outcomes, with some studies finding protection against vascular comorbidities and dementia.
One study reported that participants following a ketogenic diet experienced significant cognitive improvements alongside increased abundance of Akkermansia muciniphila, a mucin-degrading bacterium known to maintain gut barrier integrity by stimulating mucus production and strengthening epithelial tight junctions.
Is also interacts with the immune system to promote anti-inflammatory responses and regulatory T-cell activity. Reducing systemic inflammation may indirectly support cognitive function.
Studies which explored probiotics showed positive but variable effects.
Overall, there were improvements in memory, executive function and verbal fluency, and some probiotics increased GABA production and reduced amyloid-β. Some studies found significant improvements in RBANS scores in MCI patients and reduced postoperative cognitive dysfunction in surgical patients.
However, sample sizes were smaller (often 20–100 participants) and some results were not statistically significant between groups, and efficacy was likely dependent on specific strains, doses and treatment duration.
FMT showed the most dramatic individual improvements, but evidence was extremely limited.
One small preliminary study showed an MMSE increase from 13 to 18, increased microbial richness and changes in lipid-metabolism gene expression, which authors said weresuggestive of potential metabolic pathways linking microbiota alterations to neurocognitive outcomes.
“At a population level, community education, nutritional counselling during midlife, and the inclusion of microbiota-supportive diets in national guidelines could provide scalable and cost-effective means of extending cognitive healthspan and reducing the burden of dementia,” the authors wrote.