Evidence is mounting for the non-specific effects of vaccines, but the magnitude of these effects is largely unknown
It’s 1979 and Danish anthropologist Peter Aaby has taken the long flight, once again, to the tiny West African nation of Guinea-Bissau in the midst of a devastating measles outbreak.
Mothers in Guinea-Bissau know that there’s about a 50% chance that their child will die before the age of five. Measles causes 10 to 15% of these deaths.
In December that year, however, the Bandim Health Project, involving researchers such as Peter Aaby, launched the first measles vaccination campaign, and 85% of children were inoculated.
And that’s when something really unexpected happened. Instead of childhood deaths decreasing by 10-15% in line with predictions, the mortality rate plummeted by 70%.
“Seven-zero,” says Christine Stabell Benn, a professor of global health at the University of Southern Denmark, who is also involved in the Bandim Health Project.
“That’s a tremendous reduction in mortality. Measles vaccine should not reduce mortality to that large extent according to what we know about measles vaccine and its effect.”
It’s this “central mystery” that has inspired Professor Benn to travel back and forth to the former Portuguese colony for the past 25 years to study the effect of vaccines.
“I feel we have a purpose there,” she says. “It’s a quiet place to be, a calm place in comparison with many other African countries.
“It is one of the world’s poorest countries, but that’s not very apparent when you walk around in the streets. People are not malnourished and they are actually pretty happy.”
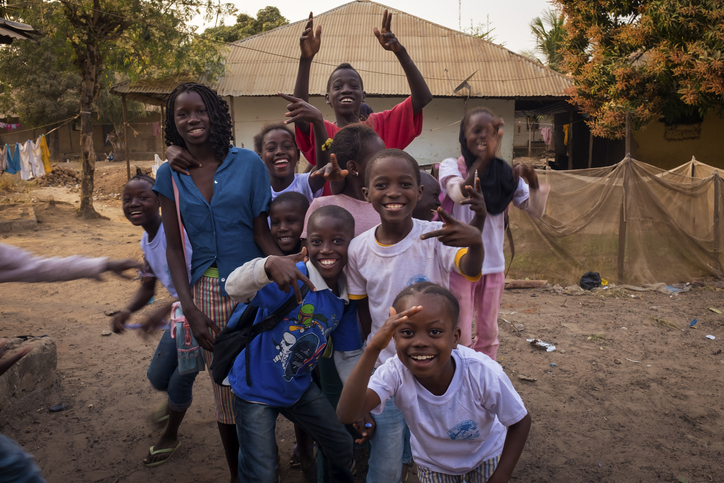
The first explanation that occurred to Professor Aaby’s team was that measles infection was somehow weakening children’s immune systems so they could not fight off other infections. That would explain why preventing the measles infection through vaccination was having such a high impact on overall mortality.
They ditched that hypothesis, however, when it became clear that unvaccinated children who had contracted a mild measles infection were actually less likely to die from other diseases.
Moreover, unvaccinated children who didn’t get the measles infection fared worse than vaccinated children. (If the measles vaccine was simply stopping measles infection, these two groups should have similar outcomes.)
So Professor Aaby proposed a radical hypothesis: the measles vaccine was protecting children against diseases other than measles.
He coined the term the “non-specific effects of vaccines” and proposed that the measles vaccine was training the immune system to recognise other pathogens.
This did not go down well.
“Stories circulated about the crazy anthropologist who was said to think that he could teach medicine something new,” says Professor Aaby. “Telephones calls were cut short, letters were not answered, and collaborators were warned against collaborating with me.
“The feelings of many physicians were expressed elegantly by a well-known UK professor in a review of our first paper on the beneficial non-specific effects of measles vaccine: ‘It would be laughable if it was not deplorable that a renowned institution like University of Copenhagen could employ a person who wrote such a paper’.”
This response did not deter the researchers; in fact, it spurred them on.
Decades later, Professor Aaby offers his thanks those who so thoroughly dismissed his early research.
“They obliged us to dig deeper and to repeat the observations to ensure that the controversial epidemiological observations were indeed reproducible,” he says.
After this initial revolutionary finding, the researchers turned their attention to other routine childhood vaccines.
In a randomised trial of around 4,000 neonates in Guinea-Bissau, the researchers found that BCG vaccination reduced neonatal deaths by more than a third.
This cannot be explained by the direct effects of the BCG vaccine against tuberculosis, as children generally didn’t die from tuberculosis in the first month of life, says Professor Benn.
“BCG reduced their risk of dying from sepsis and pneumonia – a purely non-specific effect of BCG, which had nothing to do with protection against tuberculosis,” she says.
As the research marched on a pattern started to emerge.
Live vaccines, including smallpox and polio vaccines, appeared to have beneficial non-specific effects on mortality.
But non-live vaccines, including the diphtheria, tetanus and pertussis vaccine, inactivated polio, hepatitis B and H1N1 influenza vaccines, had negative non-specific effects.
In Guinea-Bissau, children who received the DTP vaccine had a five-fold higher mortality than children who didn’t receive the vaccine.
“We have repeated this finding many times,” Professor Benn says. “Protection against diphtheria, tetanus and pertussis seems to come at a very high price: increased risk of dying from other infections, such as respiratory infections, particularly for females.”
The researchers have now completed studies on a total of four live vaccines and six non-live vaccines and the pattern is very consistent.
Curiously, it seems to be possible to reverse the negative non-specific effect of non-live vaccines by rearranging the vaccination schedule so that you end up with a live vaccine as your last vaccine, according to Professor Benn.
If it’s true that live vaccines train our immune systems to fight off a wide range of infectious diseases (and this effect is clinically significant, as all this research suggests), this has broad implications for global public health.
We may need to consider bringing back live vaccines for diseases that have already been eradicated, such as smallpox (and polio and TB in high-income countries), says Professor Benn.
Smallpox hasn’t been common since 1977, and the WHO stopped global vaccinations in 1980.
But a study of around 1,900 people in Guinea-Bissau showed that people who received the smallpox vaccine (as determined by their vaccination scar) had around 40% lower all-cause mortality than those who had never been vaccinated against smallpox, even though smallpox was not causing any deaths.
“And in Denmark, we see that those who just got the smallpox vaccine before the phase out (compared with those who didn’t get it) have a 45% lower risk of dying of natural causes up to 45 years of age,” says Professor Benn.
This finding is based on an observational study of around 48,000 schoolchildren born in Copenhagen in the 1960s and 70s.
“Nobody cared to investigate what happened when we stopped smallpox vaccine because everyone just assumed that it protected against smallpox and nothing else,” says Professor Benn.
“But, actually, it seems like it was a huge mistake to stop smallpox vaccine even in spite of the fact that it was a strong vaccine that also had serious side effects.
“We might have deprived ourselves from something really beneficial by stopping smallpox vaccine and we are afraid that something similar is about to happen with the eradication of polio and measles viruses because, from all we see, the two corresponding live vaccines, live oral polio vaccine and live measles vaccine, have strong beneficial non-specific effects.
“So, if we just eradicate the virus and stop vaccinating, again, we might actually see the paradox that the eradication leads to increased overall morbidity and mortality because we stopped the vaccines.”
COUNTER-RESEARCH
The research on the non-specific effects of vaccines in West Africa has not gone unchallenged.
Another research team in the US have harked back to Professor Aaby’s original hypothesis to explain why measles vaccination causes a larger reduction in mortality than expected.
A paper published in Science in 2015, argues that measles infection causes lasting damage to the immune system, which leaves children vulnerable to other diseases for two to three years after measles infection.
We know that the measles vaccine targets and destroys memory B and T lymphocytes. This could make the immune system forget how to fight off diseases the body has already encountered – causing “immune amnesia”, the researchers hypothesised.
Their mathematical modelling of seven decades’ worth of data from the US, UK and Denmark found that every time measles infection rates peaked, deaths rates from other diseases also increased in the subsequent 28 months.
This association held true across countries, genders, age groups and was apparent in the decades before and after mass measles vaccination.
“Our analysis certainly does not rule out other possible non-specific direct immune benefits of the vaccine shot itself,” says co-author Assistant Professor Michael Mina, who is now a pathologist at the Harvard School of Public Health.
“Rather, it is likely that both mechanisms may work hand in hand,” he says.
BACKLASH
The Mina et al paper made waves. Researchers who had spent their whole careers studying the non-specific effects of vaccines baulked at the result.
“Importantly, it is contradicted by real life data,” says Professor Benn.
For the Mina et al hypothesis to be true, individuals should be at higher risk of mortality from other infectious diseases after measles infection. However, published studies from Guinea-Bissau, Senegal, and Bangladesh confirm that there is a trend towards lower mortality in individuals who survive acute measles infection, says Professor Benn.
Moreover, measles vaccination is associated with a reduced risk of infectious disease in Denmark, where measles infection no longer occurs. This is difficult to explain unless we accept that the measles vaccination has beneficial non-specific effects.
Similar effects are seen for other live vaccines, such as oral polio vaccine, BCG and smallpox vaccine, “and they cannot be explained by prevention of long-term immune suppression by the vaccine diseases, as most of the diseases are not around anymore”, says Professor Benn.
WORLD STAGE
A few years ago, the WHO elevated the status of the Danish research by taking a serious interest in the non-specific effects of vaccines.
By this point, hundreds of peer-reviewed papers had been published on the topic over four decades. Professor Aaby sent this author a list of 329 references to his team’s research.
The WHO Strategic Advisory Group of Experts commissioned two systematic reviews to decide if there was enough evidence to consider changing vaccine schedules.
One review analysed 68 articles on the mortality rates of vaccinated versus non-vaccinated children in Africa, North America and Asia. The other review examined 77 papers on the underlying immunological mechanisms that might be driving these effects.
The first review concluded that the BCG and measles vaccines were reducing mortality more than could be explained by the targeted effects of the vaccines. So, that was good news for researchers working in this field.
The review also found that “receipt of DTP may be associated with an increase in all-cause mortality”, which was very concerning.
But the second review was inconclusive.
Putting the two 2016 papers side by side, “there is some limited evidence from an epidemiological perspective that there are non-specific effects of childhood vaccines”, says Dr Rama Kandasamy, a paediatrician at Sydney Children’s Hospital and a lecturer at UNSW who co-authored the second paper. “However, there isn’t any evidence for exactly how that may be caused. It may be purely an association.”
Without a mechanism, and in the absence of RCTs, it could just be that various forms of research bias were masquerading as a non-specific effect of the vaccines.
For instance, if sick children were less likely to get vaccinated in these trials, that could make it seem like the vaccine was more effective at reducing deaths than it actually was.
The major limitation of this field of research is its dependence on observational studies, which cannot demonstrate causation.
But this methodological approach is “absolutely essential for what we have been doing”, says Professor Aaby, because “you cannot plan to study the things you have never thought about”.
“Yes, observational studies have bias, we all know that,” he says. “But there is a very strange pattern of different vaccine effects here, which nobody has been able to explain with bias.
“Others have tried to explain away the DTP effect by saying that it is the sick children who are brought for consultation, who then gets vaccinated, creating a false association between being DTP vaccinated and higher mortality – but that explanation cannot co-exist with the observation in relation to BCG and measles vaccination (unless one presumes that these vaccines have extremely beneficial non-specific effects).”
There are obvious ethical and practical barriers to running RCTs on routine childhood vaccines. The best approach is to merge multiple lines of evidence with different underlying bias structures to build a strong argument for causality, the Danish researchers wrote in a paper last year.
Immunologists largely accept, based on animal and lab studies, that the non-specific effects of vaccines are real, says Dr Kandasamy.
“But in terms of the magnitude of those kinds of effects, that’s largely unknown,” he says. “It’s likely that they are relatively minor in the grand scheme of things.”
Other interventions, such as clean water, good nutrition and “actually just getting vaccines into kids”, are probably going to be far more important for driving down mortality than the non-specific effects of vaccines, so it’s hard to drum up interest in this area of research even among drug manufacturers, he says.
But, once we accept that vaccines probably have a much broader range of effect than we ever could have imagined, the research focus tends to shift – and it looks like this shift is happening already.

IN THE CLINIC
In Melbourne, for instance, researchers at the Murdoch Children’s Research Institute (MCRI) have launched an RCT to test whether the BCG vaccine reduces allergies or eczema in the first year of life.
The MIS BAIR (Melbourne Infant Study: BCG for Allergy and Infection Reduction) is recruiting 1,400 infants who will receive the BCG vaccine at birth.
“There is some evidence that BCG vaccination at birth protects against allergic disease,” says Professor Nigel Curtis, who is running the MIS BAIR trial.
But most of the previous studies on BCG and allergy have been underpowered, observational or conflicting, he says.
And, without a large RCT, there is insufficient evidence to recommend this intervention right now.
In other areas of practice, the off-target effects of vaccines have been used as experimental treatments already. The BCG vaccine has been trialled in the treatment of bladder cancer and malignant melanoma.
It’s thought that the bacteria in the BCG vaccine makes the innate immune system more alert to threats in general, and more likely to attack cancer cells.
The measles-mumps-rubella vaccine has also been explored as a treatment for cutaneous warts, but mainstream dermatologists tend to dismiss this as pseudoscience.
That hasn’t stopped the trials from being published. A randomised study of 150 patients published in the Journal of Clinical Aesthetic Dermatology last year claimed that injecting the MMR vaccine directly into a wart led to clearance in 70% of patients. (Warts disappeared in only 10% in the group that received saline injections in this study.)
Advocates of this treatment hypothesise that the MMR vaccine increases the immune system activity around the wart, which leads to its clearance. An RCT of 30 people in Egypt is now comparing the cryotherapy to intralesional MMR vaccine for warts.
An even weirder association has been found between the rotavirus vaccine and type 1 diabetes. An observational study by MCRI researchers recently found that the incidence of type 1 diabetes dropped 14% after the introduction of two oral rotavirus vaccines in Australia in 2007.
UNINTENDED EFFECTS
Currently, the research into the unintended effects of vaccines can be crudely divided into two areas, says Dr Jim Buttery, a paediatric infectious diseases physician and vaccinologist at Monash University.
“There are the pure non-specific effects of vaccination that we don’t fully understand,” he says. This is where the vaccine seems to make the immune system wiser and helps it defeat pathogens that are not the direct targets of the vaccine.
“The second category, if you like, is where we introduce a vaccine and we end up learning more about what the bug that we are vaccinating against actually did in the community,” he says.
“And the example of that would be when we introduced rotavirus vaccination and there was an approximately 20% drop in febrile convulsions in both Australia and the US,” he says. “We never appreciated that rotavirus had much involvement in febrile convulsions.”
There’s also a third category; where a vaccine has a beneficial, specific effect that it wasn’t designed to have.
An example of this is the serogroup B meningococcal vaccine, MeNZB, which might provide some protection against gonorrhoea.
After New Zealand rolled out MeNZB vaccinations for more than a million people, researchers analysing the retrospective data found that the vaccine was 31% effective at stopping gonorrhoea infections.
This was big news because we’ve never been able to develop a vaccine against gonorrhoea specifically.
The 2017 New Zealand study on people aged 15 to 30 years was observational, so it doesn’t provide causality.
However, research by Associate Professor Kate Seib, a microbiologist at Griffith University in Queensland, published last year, revealed a plausible biological mechanism by which MeNZB and the newer MenB vaccine Bexsero could be shielding people against gonorrhoea.
Both bacteria are similar in terms of their protein makeup, so it makes sense that the MenB vaccine might provide cross-protection against gonorrhoea, says Professor Seib.
If RCTs confirm the result, there would be two options: we could use the MenB vaccine to directly vaccinate against gonorrhoea, or we could use our improved understanding of the protective immune response to gonorrhoea to develop a more potent, specific gonorrhoea vaccine, says Professor Seib.
GSK, which manufactures Bexsero, funded the New Zealand trial but wasn’t involved in Professor Seib’s study.
It’s not unusual for vaccines to offer cross-protection. The group A meningococcal conjugate vaccine also protects against tetanus, for example.
This is because the vaccine uses a tetanus toxoid as a carrier protein, which induces an immune response against tetanus.
Neonatal cases of tetanus dropped 25% across sub-Saharan Africa in the 2010s, when the “MenAfriVac” program kicked off.
ATTACK OF THE ANTI-VAXERS
Research into the non-specific effects of vaccines has always been difficult to play in the media because it complicates the core public health message about vaccines having straightforward, safe, targeted effects – and any findings of harmful off-target effects can be pounced on by anti-vaxers.
“I spend a lot of time speculating about how to communicate these findings in a manner that doesn’t create panic about vaccines,” says Professor Benn.
“I’m so worried about creating a general sense that vaccines are unsafe, because that is definitely not my agenda,” she says.
“On the other hand, I’m concerned that the health authorities are actually not responding to these findings, so I feel the need to raise my voice.”
It’s becoming increasingly clear that vaccines aren’t “one thing”; each vaccine interacts with and trains the immune system in a different way, she says.
We should just talk about each and every vaccine by their name and attribute the benefits and the potential harms to each one of them, she says.
“That would be such a good starting point for a more nuanced debate about vaccines.”


